…
Action Limits and Alert Levels
Action limits and alert levels are used, for example, as criteria to control microbial or suspended particle limits.
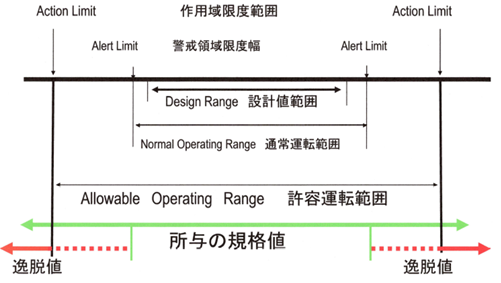
The action limit, or action range limit, is the limit of the permissible operating range, which, if exceeded, constitutes a deviation. In such cases, appropriate investigation and corrective measures based on the investigation must be implemented.
The Alert level is the alert area limit width, within which the normal operating range is located.
Alert levels are early warnings of potential deviations from normal operating conditions and validated states. Exceeding an alert level does not necessarily require corrective action, but it does require appropriate scrutiny and follow-up, or preventive action, to address potential problems.
Alert levels are set based on periodic eligibility validation trend data and are reviewed periodically. Alert levels can be based on a number of parameters, such as adverse trends, individual transient deviations (excursions) that exceed set limits, recurring events, etc.
For example, consider the temperature control of a refrigerator (2°C to 8°C). In most cases, the temperature setting of a refrigerator is supposed to be 5°C. This is to prevent the temperature from temporarily rising or falling and deviating when the door is opened. This is to prevent the temperature from temporarily rising or falling and deviating when the door is opened.
Let’s say the alert level is set between 3°C and 7°C. In this case, the alert level would be set to 5°C again when the alert level is exceeded or when the trend is moving in the direction of deviation. This is preventive action.
If the temperature exceeds the alert level or is heading in the direction of deviation, a problem may be lurking somewhere.
For example, the refrigerator door may no longer close properly, there may be a gap, or the door may be opened and closed too often.
Since it is not yet a deviation, no corrective action is required, but preventive action is needed to avoid deviation.
transient deviation(Excursion)and temporary deviations(Spike)
A transient deviation is a deviation of relatively short duration (e.g., 20 minutes or less).
Also, a temporary deviation is a deviation only for a brief moment (e.g., one minute).
Some companies allow transient or primary deviations and do not take them up as deviations unless they exceed the prescribed time.
However, alert-level transient deviations should be documented and reviewed and should include an investigation to determine if the transient deviation is a single (isolated) event or if the result indicates a bad trend or system degradation.
This means that each action limit transitory deviation should be investigated to determine possible root causes and potential impacts on product quality and manufacturing processes.
related product

]]>
Related Articles
- Action Limits and Alert Levels in Pharmaceutical Quality Systems
- Preventive Action as Risk Management: Understanding CAPA in Pharmaceutical and Medical Device Industries
- Understanding FDA Advisory Action Letters: Warning Letters and Untitled Letters
- Understanding the Differences Between Correction, Corrective Action, and Preventive Action
- Three Critical Actions That Destroy Audit Trails: Major Risks in the Pharmaceutical Industry
- Why Horizontal Deployment is Not Preventive Action: Understanding CAPA Fundamentals


Comment